Description
NAD+ 1000mg (Nicotinamide Adenine Dinucleotide) – Research Use Only (RUO)
Product Specification
- Product Name : NAD+ (Nicotinamide Adenine Dinucleotide)
- CAS Number : 53-84-9
- Synonyms : DPN, Coenzyme 1, NAD Plus
- Mass per Unit : 1000mg (Lyophilized Powder)
- Molecular Formula : C21H27N7O14P2
- Molecular Weight : 663.43 g/mol 6
- Purity : ≥99% (HPLC Verified)
- Form : Lyophilized White Powder
- Endotoxin Level : Less than 1.0 EU/mg
Disclaimer : For Research Use Only (RUO). Not for human use.
NAD+ 1000mg is a classical metabolic coenzyme for many redox reactions and also supports NAD-consuming signaling enzymes such as sirtuins and PARPs. In bench workflows, NAD+ is typically selected when the experimental design requires the final substrate rather than relying on precursor conversion steps.
Chemical Identity & Mechanism
What NAD+ 1000mg Does in Research Systems
Redox Cofactor : NAD+ 1000mg participates directly in electron transfer (NAD+/NADH), supporting core metabolic enzyme assays and bioenergetic models.
Consumable Substrate : NAD+ 1000mg is consumed by NAD-dependent enzymes (notably Sirtuins and PARPs), linking NAD+ availability to enzyme kinetics and signaling readouts in laboratory models.
Why “Final Molecule” Matters : Precursor inputs (NR/NMN) must be converted through cellular pathways NRK → NMN → NMNAT → NAD+ and this conversion can be rate-limited or context-dependent in experimental systems.
Chemical structure of NAD+ 1000mg
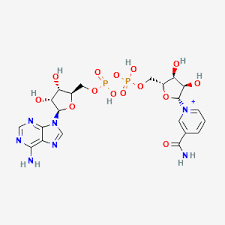
Verified 2D chemical structure of NAD+ 1000mg (Nicotinamide Adenine Dinucleotide) a dinucleotide coenzyme central to cellular redox chemistry and NAD+ dependent enzyme pathways.
NAD+ 1000mg (nicotinamide adenine dinucleotide) chemical structure showing the adenine and nicotinamide rings with two phosphate groups; molecular formula C21H27N7O14P2 NAD Plus research grade RUO
Comparative Research Context
Direct NAD+ vs Precursors vs Incretin-Class Research Tools
| Feature | NAD+ (Direct) | NR / NMN (Precursors) | Reta / Tirz / Sema |
|---|---|---|---|
| Final Molecule | Yes | No | Not applicable |
| Direct Redox Activity | Yes | No | Not applicable |
| Conversion Requirement | None | Requires enzymatic conversion | Receptor-mediated class |
| Assay Availability | Direct substrate; immediate use | Salvage-pathway dependent | Pathway-specific signaling |
| Form Stability | Sensitive once in solution; integrity checks required | Generally higher stability | Product-specific controls |
| Primary Use Context | Bioenergetics, redox & enzyme assays | Precursor flux studies | Metabolic receptor research |
Practical Interpretation for Researchers
- Direct NAD+ 1000mg is selected when the experiment requires an immediately available cofactor without reliance on conversion steps.
- NR/NMN are used when studying precursor pathways and salvage kinetics.
- Reta/Tirz/Sema represent a different mechanistic class focused on receptor-level pharmacology rather than NAD biochemistry.
Rsearch context and market status
1) Pathway clarity (what researchers actually agree on)
Pathway clarity (what researchers actually agree on)
Most published approaches to increasing cellular NAD focus on precursor conversion pathways (NR/NMN AD+) and intracellular recycling mechanisms. Recent human metabolic studies comparing different NAD+ 1000mg boosting strategies show that precursor forms and NAD+ 1000mg metabolism can influence circulatory NAD+ levels and downstream metabolic pathways, reinforcing the mechanistic reality that precursor conversion and salvage context are central to NAD+ availability.
Christen et al., “Differential impact of NAD+ 1000mg boosters on circulatory NAD and metabolism,” Nature Metabolism, 2026.
Longer-horizon evidence remains actively studied; a controlled safety/metabolism study of prolonged NR supplementation has been published, reinforcing that mechanistic claims must stay evidence-based and population-specific.
2) Degradation relevance (why stability is not “cosmetic”)
NAD+ 1000mg in solution can undergo degradation, and laboratory studies have shown that even small changes in buffer composition, temperature, and storage conditions can affect the stability of NAD+ and related nicotinamide cofactors; this highlights the importance of monitoring solution integrity in experimental workflows. The concern is not appearance alone degradation can shift the chemical profile and confound pathway readouts, especially in enzyme-linked assays and signaling studies.
Wolfe, K.D. et al. (2024) – “Long-Term Stability of Nicotinamide Cofactors in Common Biocatalytic Buffers,” showing how NAD+ and NADH stability varies with buffer and temperature and how degradation can be measured and monitored in lab settings.
3) Regulatory Caution
FDA communications state that NAD+ drug products are not FDA-approved and caution against the use of food-grade NAD+ materials where pharmaceutical-level quality controls are required due to contamination and endotoxin risk. This is why RUO suppliers must remain strictly research-positioned, supported by lot-specific COA and analytical verification. FDA Compounding Safety Communication, 2025.
Why Researchers Source NAD+ 1000mg from Profound Aminos
Quality and analytical documentation are the primary differentiators in the RUO market, where contamination, hydrolytic degradation, and mislabeled purity can directly compromise experimental outcomes. Profound Aminos maintains an institutional-grade supply chain designed specifically for the rigorous requirements of USA-based laboratory research.
- Lot-Specific Transparency (COA-First) : We prioritize an identity- and purity-first workflow. Every compound is batch-tracked and accompanied by a lot-specific Certificate of Analysis (COA), featuring HPLC chromatograms and Mass Spectrometry data to verify purity levels of ≥99%.
- USA-Based Cold-Chain Logistics : Chemical stability is critical for research integrity. To prevent thermal and hydrolytic degradation, all lyophilized inventory is stored at -20°C in our USA facility and shipped under strict cold-chain protocols to ensure immediate experimental potency upon arrival.
- Endotoxin Monitoring & Safety : To ensure suitability for sensitive in vitro assays and cellular modeling, we screen our products for bacterial endotoxins, maintaining levels at <1.0 EU/mg. This eliminates the “grey-market” risk of pyrogenic reactions that can skew research data.
- Contaminant Risk Awareness : We actively educate the research community about the risks associated with “food-grade” or improperly labeled “research chemicals”. By providing pharmaceutical-grade APIs, we mitigate the risk of unknown byproducts, such as trifluoroacetic acid (TFA) or heavy metals, which are common in unregulated supply chains.
Hangling & Stability
Lyophilized powder : longer stability when stored cold; shipping at ambient may be acceptable short-term if stored cold on arrival.
Reconstituted solutions : degrade faster; refrigeration is commonly treated as mandatory, and solutions are time-limited.
Integrity check : clear vs yellowing/precipitate is used as a lab flag for possible degradation.
NAD+ 1000mg Research (RUO)
Frequently Asked Questions
Q1) Is degraded NAD+ a problem in assays?
Yes. If NAD+ degrades in solution, the resulting chemical profile can alter enzyme-substrate availability and confound NAD+-linked readouts. Visual integrity flags (yellowing/precipitate) are commonly treated as a warning sign in lab handling.
Q2) What distinguishes NAD+ from NR/NMN in models?
NR and NMN are precursor molecules that must undergo enzymatic conversion before becoming NAD+, whereas NAD+ is the final substrate used directly in redox and NAD-dependent enzyme assays. This distinction is relevant in experimental design because precursor pathways can introduce time- and enzyme-dependent variability, while direct NAD+ provides an immediately available cofactor for controlled laboratory workflows.
Q3) How is stability monitored in practice?
For RUO handling, solution clarity is used as a practical integrity check: clear is treated as acceptable; yellowing/precipitate is treated as potential degradation.
Q4) What drove FDA warnings relevant to NAD+ products?
FDA communications have cautioned that NAD+ drug products are not FDA-approved and have raised concerns when food-grade NAD+ materials are used in settings requiring appropriate quality controls due to contamination and endotoxin risk. This reinforces the need for verified quality systems and RUO-only positioning.
Q5) Does NAD+ belong to the incretin category (Reta/Tirz/Sema)?
No. NAD+ is a coenzyme/substrate in redox and NAD+-dependent enzyme pathways, while Reta/Tirz/Sema are receptor-acting incretin mimetics (GLP-1/GIP class).
Q6) Why do some labs prefer direct NAD+ instead of only precursors?
Because NAD+ is the final substrate, it can be used to probe pathway behavior without relying solely on conversion steps—useful in assay development and controlled mechanistic testing.
Q7) What is the biggest RUO compliance mistake in this category?
Using RUO positioning while implying therapeutic intent. FDA enforcement has targeted sellers who label products RUO while marketing for human use.
Q8) What market confusion should the page address (to build trust)?
Consumers often do not understand the difference between RUO vendors, prescription compounding, and dietary supplements—clear education reduces risk and improves credibility.
Q9) What does “1000mg” actually mean for a research buyer?
It typically indicates total mass per unit for reagent planning, stability testing, and multi-assay workflows—verify exact content and purity per lot using COA.
Q10) What’s the safest Google-policy stance for NAD+ content?
Keep content factual, cite sources, avoid disease/therapeutic claims, and clearly state RUO status while educating on regulatory context.








Reviews
There are no reviews yet.